P23938
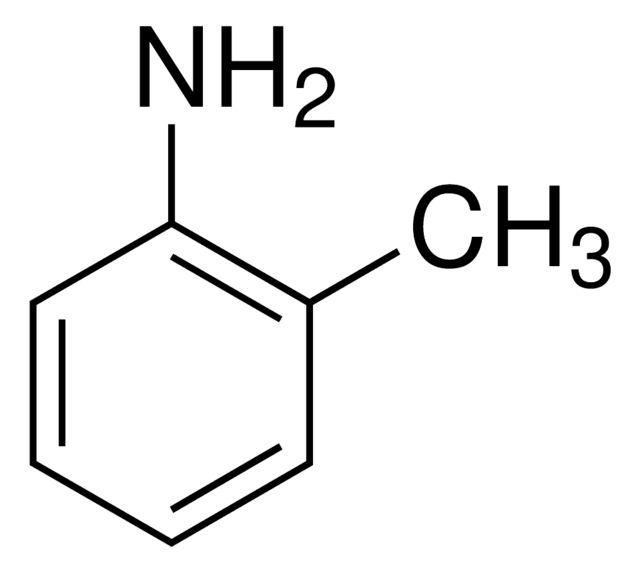
o-Phenylenediamine
flaked, 99.5%
Manufacturer: Sigma Aldrich
CAS Number: 95-54-5
Synonym(S): 1,2-Diaminobenzene, 1,2-Phenylenediamine, OPD
Select a Size
| Pack Size | SKU | Availability | Price |
|---|---|---|---|
| 5 G | P23938-5-G | In Stock | ₹ 3,453.18 |
| 100 G | P23938-100-G | In Stock | ₹ 5,152.70 |
| 500 G | P23938-500-G | In Stock | ₹ 7,003.78 |
P23938 - 5 G
In Stock
Quantity
1
Base Price: ₹ 3,453.18
GST (18%): ₹ 621.572
Total Price: ₹ 4,074.752
vapor density
3.7 (vs air)
Quality Level
200
vapor pressure
0.01 mmHg ( 25 °C)
Assay
99.5%
form
powder
bp
256-258 °C
mp
100-102 °C
SMILES string
Nc1ccccc1N
InChI
1S/C6H8N2/c7-5-3-1-2-4-6(5)8/h1-4H,7-8H2
InChI key
GEYOCULIXLDCMW-UHFFFAOYSA-N
Other Options
| Image | Product Name | Manufacturer | Price Range | |
|---|---|---|---|---|
Sigma Aldrich Fine Chemicals Biosciences o-Phenylenediamine tablet, 20 mg substrate per tablet | 95-54-5 | MFCD00007721 | 50TAB | -- | ₹ 29,328.17 | ||
Sigma Aldrich Fine Chemicals Biosciences o-Phenylenediamine Peroxidase substrate, >=98.0%, powder | 95-54-5 | MFCD00007721 | 50G | -- | ₹ 26,057.42 | ||
Sigma Aldrich Fine Chemicals Biosciences o-Phenylenediamine flaked, 99.5% | 95-54-5 | MFCD00007721 | 5G | -- | ₹ 4,939.50 | ||
o-Phenylenediamine | -- | ₹ 6,246.03 | ||
1,2-Phenylenediamine | -- | ₹ 10,580.00 | ||
o-Phenylenediamine | -- | ₹ 2,533.05 | ||
o-Phenylenediamine | -- | ₹ 25,395.45 - ₹ 42,314.93 | ||
o-Phenylenediamine | -- | ₹ 29,130.08 - ₹ 45,151.08 | ||
95-54-5 | O-Phenylenediamine | -- | ₹ 1,513.00 - ₹ 15,575.00 |
Description
- Application: o-Phenylenediamine can be used: To prepare quinoxalines, benzimidazoles, and 1H-1,5-benzodiazepines by condensing with aryl aldehydes or ketones.[1] In the H2O mediated one-pot preparation of 1,2-disubstituted benzimidazoles by reacting with aldehydes in the presence of trimethylsilyl chloride.[2]In the copper-catalyzed synthesis of alkyne substituted quinoxalines by reacting with terminal alkynes.[3]In the preparation of 1,5-benzodiazepine derivatives by reacting with different ketones under neat reaction conditions via ytterbium trichloride catalyzed condensation reaction.[4]
SAFETY INFORMATION
Pictograms
Signal Word
Danger
Hazard Statements
Precautionary Statements
P273 - P280 - P301 + P310 - P302 + P352 + P312 - P304 + P340 + P312 - P305 + P351 + P338
Hazard Classifications
Acute Tox. 3 Oral - Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Aquatic Acute 1 - Aquatic Chronic 1 - Carc. 2 - Eye Irrit. 2 - Muta. 2 - Skin Sens. 1
WGK
WGK 3
Flash Point(F)
276.8 °F - closed cup
Flash Point(C)
136 °C - closed cup