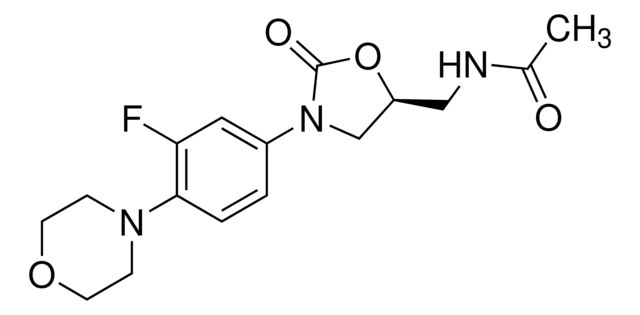
Valsartan Related Compound B
Pharmaceutical Secondary Standard; Certified Reference Material
Manufacturer: Supelco
CAS Number: 952652-79-8
Synonym(S): N-(1-Oxobutyl)-N-[[2′-(2H-tetrazol-5-yl)[1,1′-biphenyl]-4-yl]methyl]-L-valine, N-butyryl-N-{[2′-(1H-tetrazole-5-yl)-biphenyl-4-yl]methyl}-L-valine, (S)-N-Butyryl-N-{[2′-(1-H-tetrazole-5-yl)-biphenyl-4-yl]methyl}valine
Select a Size
| Pack Size | SKU | Availability | Price |
|---|---|---|---|
| 30 MG | PHR1876-30-MG | In Stock | ₹ 72,679.05 |
PHR1876 - 30 MG
In Stock
Quantity
1
Base Price: ₹ 72,679.05
GST (18%): ₹ 13,082.229
Total Price: ₹ 85,761.279
grade
certified reference materialpharmaceutical secondary standard
Quality Level
300
Agency
traceable to USP 1708784
API family
valsartan
CofA
current certificate can be downloaded
packaging
pkg of 30 mg
application(s)
pharmaceutical
format
neat
storage temp.
2-8°C
SMILES string
CCCC(=O)N(Cc1ccc(cc1)-c2ccccc2-c3nnn[nH]3)[C@@H](C(C)C)C(O)=O
Other Options
| Image | Product Name | Manufacturer | Price Range | |
|---|---|---|---|---|
Valsartan Related Compound B Pharmaceutical Secondary Standard, MilliporeSigma™ Supelco™ | -- | ₹ 79,032.00 | ||
Valsartan Related Compound B | -- | ₹ 1,78,331.05 | ||
952652-79-8 | (S)-2-(N-((2'-(1H-Tetrazol-5-yl)-[1,1'-biphenyl]-4-yl)methyl)butyramido)-3-methylbutanoic acid | -- | ₹ 10,858.00 - ₹ 85,618.00 |
Related Products
Description
- General description: This Certified Reference Material (CRM) is produced and certified in accordance with ISO 17034 and ISO/IEC 17025. All information regarding the use of this CRM can be found on the certificate of analysis.Pharmaceutical secondary standards for application in quality control provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.It is an impurity of the potent, highly selective, and orally active antagonist of the angiotensin II AT1-receptor, valsartan, used widely for the treatment of hypertension.
- Application: This pharmaceutical secondary standard can also be used as follows:Development of a reversed-phase high-performance liquid chromatographic (RP-HPLC) method for the determination of valsartan and its related impurities in pharmaceutical dosage formsImpurity testing of valsartan, amlodipine besylate, and hydrochlorothiazide in their combined dosage form by a stability-indicating ultra-high performance liquid chromatography (UHPLC)Simultaneous determination of amlodipine and valsartan in their combined dosage form, in the presence of their degradation products by a gradient reversed phase-liquid chromatographic (RP-LC) method[1]Separation and detection of nitrosamines and other related impurities in valsartan and losartan using supercritical fluid chromatography (SFC) in a single runDevelopment and validation of a UHPLC method for the estimation of sacubitril, valsartan, and their related impurities in their combined dosage form, following ICH Q2 (R1) guideline[2]
- Analysis Note: These secondary standards offer multi-traceability to the USP, EP and BP primary standards, where they are available.
- Footnote: To see an example of a Certificate of Analysis for this material enter LRAB1042 in the Documents slot below. This is an example certificate only and may not be the lot that you receive.
SAFETY INFORMATION
Pictograms
Signal Word
Warning
Hazard Statements
Precautionary Statements
P202 - P261 - P271 - P280 - P304 + P340 + P312 - P308 + P313
Hazard Classifications
Repr. 2 - STOT SE 3
Target Organs
Central nervous system
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable