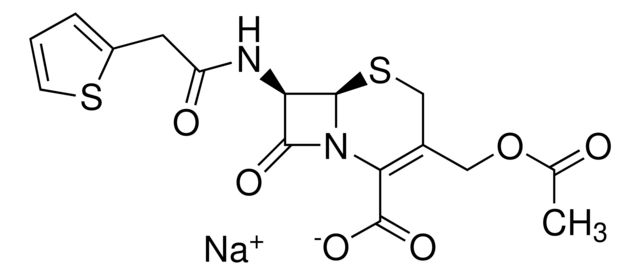
Cefalotin sodium
European Pharmacopoeia (EP) Reference Standard
Manufacturer: Sigma Aldrich
CAS Number: 58-71-9
Synonym(S): Cephalothin sodium salt, 7-(2-Thienylacetamido)cephalosporanic acid sodium salt, Cephalotin sodium salt
Select a Size
| Pack Size | SKU | Availability | Price |
|---|---|---|---|
| 170 MG | C0682000-170-MG | In Stock | ₹ 14,267.35 |
C0682000 - 170 MG
In Stock
Quantity
1
Base Price: ₹ 14,267.35
GST (18%): ₹ 2,568.123
Total Price: ₹ 16,835.473
grade
pharmaceutical primary standard
API family
cefalotin
manufacturer/tradename
EDQM
application(s)
pharmaceutical (small molecule)
format
neat
storage temp.
2-8°C
SMILES string
[Na+].CC(=O)OCC1=C(N2[C@H](SC1)[C@H](NC(=O)Cc3cccs3)C2=O)C([O-])=O
InChI
1S/C16H16N2O6S2.Na/c1-8(19)24-6-9-7-26-15-12(14(21)18(15)13(9)16(22)23)17-11(20)5-10-3-2-4-25-10;/h2-4,12,15H,5-7H2,1H3,(H,17,20)(H,22,23);/q;+1/p-1/t12-,15-;/m1./s1
InChI key
VUFGUVLLDPOSBC-XRZFDKQNSA-M
Other Options
| Image | Product Name | Manufacturer | Price Range | |
|---|---|---|---|---|
Cefalotin Sodium, British Pharmacopoeia (BP) Reference Standard, MilliporeSigma™ Supelco™ | -- | ₹ 27,946.00 | ||
Sigma Aldrich Fine Chemicals Biosciences Cephalothin Sodium Salt | 58-71-9 | MFCD00072025 | 5g | -- | ₹ 52,759.20 | ||
Chem-Impex International, Inc. Cephalothin sodium salt | 58-71-9 | MFCD00072025 | 1G | -- | ₹ 4,628.00 | ||
Chem-Impex International, Inc. Cephalothin sodium salt | 58-71-9 | MFCD00072025 | 5G | -- | ₹ 19,224.00 | ||
Cefalotin sodium | -- | ₹ 24,767.60 | ||
Cefalotin for impurity B identification | -- | ₹ 14,072.50 | ||
Cephalothin sodium salt | -- | ₹ 4,016.08 - ₹ 31,370.85 | ||
Cephalothin sodium salt | -- | ₹ 4,016.08 - ₹ 31,370.85 |
Related Products
Description
- General description: This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including SDS and any product information leaflets have been developed and issued under the Authority of the issuing Pharmacopoeia.For further information and support please go to the website of the issuing Pharmacopoeia.
- Application: Cefalotin sodium EP Reference standard, intended for use in laboratory tests only as specifically prescribed in the European Pharmacopoeia.
- Biochem/physiol Actions: Mode of Action: Disrupts the synthesis of the peptidoglycan layer of bacterial cell walls. Antimicrobial spectrum: Effective aginast both Gram-positive and Gram-negative bacteria. Mode of Resistance: Production of cephalosporinase will inactivate the product.
- Packaging: The product is delivered as supplied by the issuing Pharmacopoeia. For the current unit quantity, please visit the EDQM reference substance catalogue.
- Other Notes: Sales restrictions may apply.
SAFETY INFORMATION
Pictograms
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Resp. Sens. 1 - Skin Sens. 1
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable