ALD00352
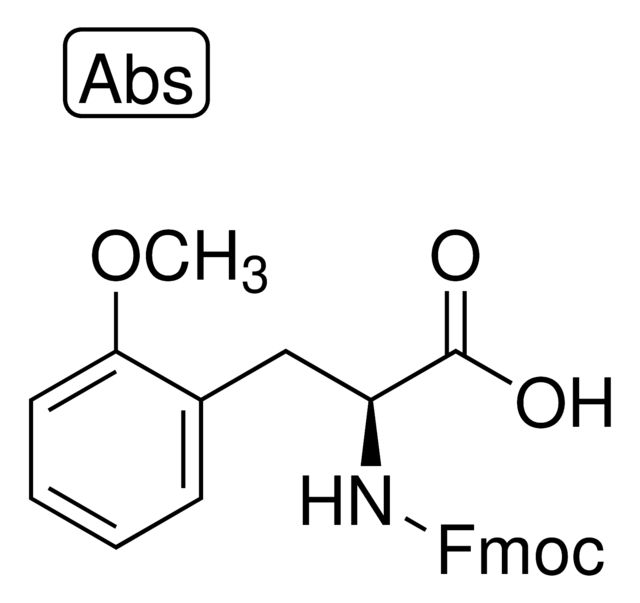
Boc-2-methoxy-L-phenylalanine
97%
Manufacturer: Sigma Aldrich
CAS Number: 143415-63-8
Synonym(S): (S)-2-(tert-butoxycarbonylamino)-3-(2-methoxyphenyl)propanoic acid
Select a Size
| Pack Size | SKU | Availability | Price |
|---|---|---|---|
| 1 G | ALD00352-1-G | In Stock | ₹ 18,629.83 |
ALD00352 - 1 G
In Stock
Quantity
1
Base Price: ₹ 18,629.83
GST (18%): ₹ 3,353.369
Total Price: ₹ 21,983.199
Quality Level
100
Assay
97%
form
powder
reaction suitability
reaction type: Boc solid-phase peptide synthesisreagent type: ligandreaction type: C-H Activation
mp
157 °C
application(s)
peptide synthesis
storage temp.
2-8°C
SMILES string
OC([C@@H](NC(OC(C)(C)C)=O)CC1=CC=CC=C1OC)=O
InChI
1S/C15H21NO5/c1-15(2,3)21-14(19)16-11(13(17)18)9-10-7-5-6-8-12(10)20-4/h5-8,11H,9H2,1-4H3,(H,16,19)(H,17,18)/t11-/m0/s1
InChI key
QMHKMTAKTUUKEK-NSHDSACASA-N
Other Options
| Image | Product Name | Manufacturer | Price Range | |
|---|---|---|---|---|
eMolecules BOC-2-METHOXY-L-PHENYLALANINE | 143415-63-8 | MFCD01860640 | 1g | -- | ₹ 54,194.77 | ||
Sigma Aldrich Fine Chemicals Biosciences Boc-2-methoxy-L-phenylalanine 97% | 143415-63-8 | MFCD01860640 | 1G | -- | ₹ 30,527.00 | ||
Boc-2-methoxy-L-phenylalanine | -- | ₹ 8,989.00 - ₹ 40,495.00 | ||
143415-63-8 | (S)-2-((tert-Butoxycarbonyl)amino)-3-(2-methoxyphenyl)propanoic acid | -- | ₹ 3,560.00 - ₹ 73,336.00 |
Description
- Application: Unnatural amino acid was derived from a C-H Activation methodology developed by Jin-Quan Yu and coworkers. The Pd-mediated C-C bond formation can be made in tandem with 2-Methylpyridine (Aldrich 109835).
- Other Notes: Ligand-Controlled C(sp3)–H Arylation and Olefination in Synthesis of Unnatural Chiral α–Amino Acids
SAFETY INFORMATION
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable