187240
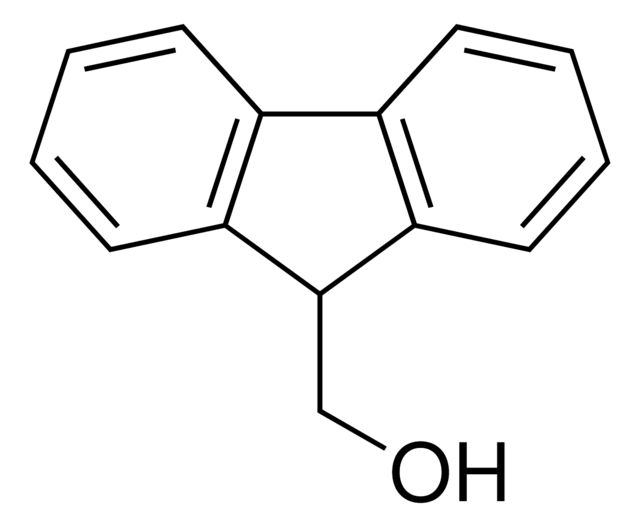
9-Anthracenemethanol
97%
Manufacturer: Sigma Aldrich
CAS Number: 1468-95-7
Synonym(S): 9-(Hydroxymethyl)anthracene
Select a Size
| Pack Size | SKU | Availability | Price |
|---|---|---|---|
| 5 G | 187240-5-G | In Stock | ₹ 3,020.00 |
| 10 G | 187240-10-G | In Stock | ₹ 6,213.55 |
| 50 G | 187240-50-G | In Stock | ₹ 27,419.73 |
187240 - 5 G
In Stock
Quantity
1
Base Price: ₹ 3,020.00
GST (18%): ₹ 543.60
Total Price: ₹ 3,563.60
Quality Level
100
Assay
97%
solubility
chloroform: soluble 20 mg/mL, clear to slightly hazy, light yellow to yellow
SMILES string
OCc1c2ccccc2cc3ccccc13
InChI
1S/C15H12O/c16-10-15-13-7-3-1-5-11(13)9-12-6-2-4-8-14(12)15/h1-9,16H,10H2
InChI key
JCJNNHDZTLRSGN-UHFFFAOYSA-N
Other Options
| Image | Product Name | Manufacturer | Price Range | |
|---|---|---|---|---|
Sigma Aldrich Fine Chemicals Biosciences 9-Anthracenemethanol 97% | 1468-95-7 | MFCD00001264 | 50G | -- | ₹ 50,943.60 | ||
Sigma Aldrich Fine Chemicals Biosciences 9-Anthracenemethanol 97% | 1468-95-7 | MFCD00001264 | 5G | -- | ₹ 9,529.23 | ||
9-Anthracenemethanol | -- | ₹ 534.00 - ₹ 19,580.00 | ||
1468-95-7 | 9-Anthracenemethanol | -- | ₹ 445.00 - ₹ 3,382.00 |
Related Products
Description
- General description: 9-Anthracenemethanol participates in ring-opening polymerization of L-lactide catalyzed by alumoxane[1]. It undergoes proton exchange reaction with potassium tert-butoxide to yield potassium 9-anthracenemethoxide[2].9-Anthracenemethanol is a derivative of anthracene used in the Diels-Alder reactions. [3]
- Application: 9-Anthracenemethanol can be used:As a starting material to prepare 9-anthracenylmethyl-1-piperazinecarboxylate, which acts as a reagent in the determination of isocyanates using HPLC.[4]In the Diels-Alder reaction with dimethylacetylene-dicarboxylate to yield lactone derivatives.[5]As an initiator in the ring-opening polymerization of δ-valerolactone to yield poly(δ-valerolactone).[6]As a starting material in the synthesis of polymer-supported anthracene, which acts as a dienophile scavenger in cycloaddition reactions.[7]
SAFETY INFORMATION
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves