345350
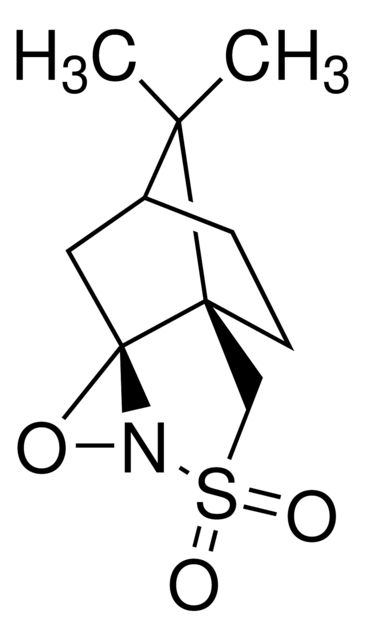
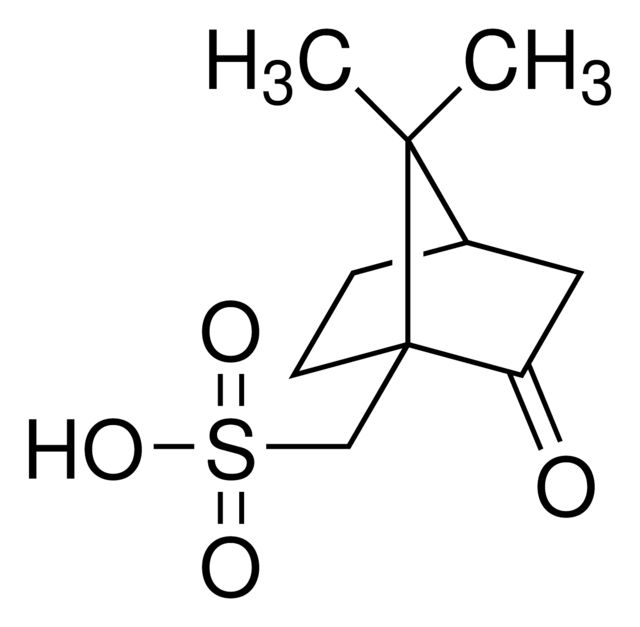
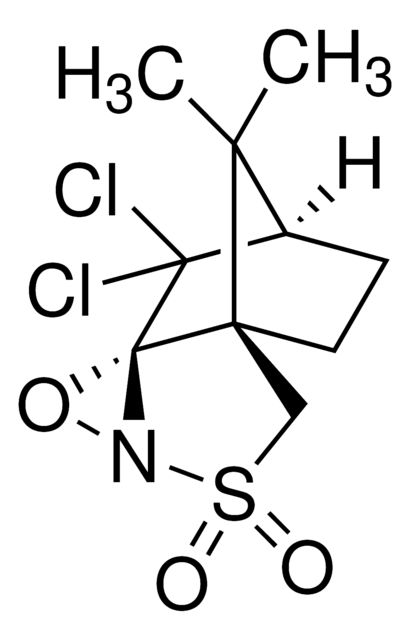
(1S)-(+)-(10-Camphorsulfonyl)oxaziridine
Manufacturer: Sigma Aldrich
CAS Number: 104322-63-6
Synonym(S): (1S)-(+)-(Camphorylsulfonyl)oxaziridine, (1S)-(+)-2,N-Epoxy-exo-10,2-bornanesultam
Select a Size
| Pack Size | SKU | Availability | Price |
|---|---|---|---|
| 1 G | 345350-1-G | In Stock | ₹ 6,679.03 |
345350 - 1 G
In Stock
Quantity
1
Base Price: ₹ 6,679.03
GST (18%): ₹ 1,202.225
Total Price: ₹ 7,881.255
form
solid
Quality Level
200
optical activity
[α]28/D +45°, c = 2 in chloroform
impurities
<1% toluene
mp
172-174 °C (lit.)
storage temp.
2-8°C
SMILES string
CC1(C)[C@@H]2CC[C@]13CS(=O)(=O)N4O[C@@]34C2
InChI
1S/C10H15NO3S/c1-8(2)7-3-4-9(8)6-15(12,13)11-10(9,5-7)14-11/h7H,3-6H2,1-2H3/t7-,9+,10+,11?/m1/s1
InChI key
GBBJBUGPGFNISJ-YDQXZVTASA-N
Other Options
| Image | Product Name | Manufacturer | Price Range | |
|---|---|---|---|---|
104322-63-6 | (1S)-(+)-(10-Camphorsulfonyl)oxaziridine | -- | ₹ 445.00 - ₹ 9,701.00 |
Related Products
Description
- Application: (1S)-(+)-(10-Camphorsulfonyl)oxaziridine can be used:To convert prochiral ketone enolates into optically active α-hydroxy ketones via enantioselective asymmetric oxidation.[1]In the synthesis of thymidine oligonucleotides connected through pyrophosphates.[2]In the asymmetric synthesis of proton pump inhibitors like (R)-Rabeprazole sodium and (R)-Lansoprazole sodium from the corresponding DBU salt of prochiral sulfide.[3]In the preparation of phosphonoacetate and thiophosphonoacetate oligodeoxynucleotides by oxidizing the corresponding phosphinoacetate.[4]
SAFETY INFORMATION
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves