663107
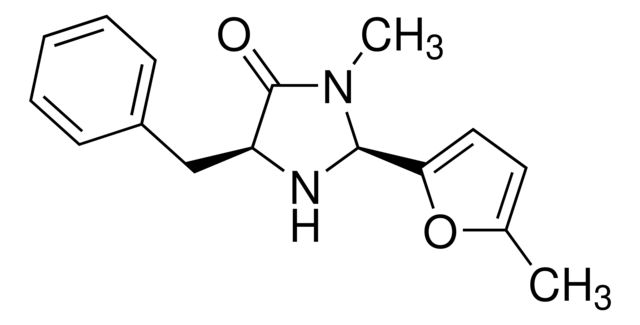
(2S,5S)-(−)-2-tert-Butyl-3-methyl-5-benzyl-4-imidazolidinone
97%
Manufacturer: Sigma Aldrich
CAS Number: 346440-54-8
Synonym(S): (2S,5S)-2-tert-Butyl-3-methyl-5-phenylmethyl-4-imidazolidinone, (2S,5S)-5-Benzyl-2-tert-butyl-3-methyl-4-imidazolidinone
Select a Size
| Pack Size | SKU | Availability | Price |
|---|---|---|---|
| 500 MG | 663107-500-MG | In Stock | ₹ 19,593.25 |
| 1 G | 663107-1-G | In Stock | ₹ 28,686.25 |
663107 - 500 MG
In Stock
Quantity
1
Base Price: ₹ 19,593.25
GST (18%): ₹ 3,526.785
Total Price: ₹ 23,120.035
Quality Level
100
Assay
97%
form
solid
mp
93-100 °C (lit.)
SMILES string
CN1[C@H](N[C@@H](Cc2ccccc2)C1=O)C(C)(C)C
InChI
1S/C15H22N2O/c1-15(2,3)14-16-12(13(18)17(14)4)10-11-8-6-5-7-9-11/h5-9,12,14,16H,10H2,1-4H3/t12-,14-/m0/s1
InChI key
SKHPYKHVYFTIOI-JSGCOSHPSA-N
Other Options
| Image | Product Name | Manufacturer | Price Range | |
|---|---|---|---|---|
Sigma Aldrich Fine Chemicals Biosciences (2S,5S)-(-)-2-tert-Butyl-3-methyl-5-benzyl-4-imidazolidinone 97% | 346440-54-8 | MFCD03426982 | 1G | -- | ₹ 45,817.20 | ||
Sigma Aldrich Fine Chemicals Biosciences (2S,5S)-(-)-2-tert-Butyl-3-methyl-5-benzyl-4-imidazolidinone 97% | 346440-54-8 | MFCD03426982 | 500MG | -- | ₹ 27,147.67 | ||
(2S,5S)-(-)-2-TERT-BUTYL-3-METHYL-5-BENZYL-4-IMIDAZOLIDINONE | -- | ₹ 2,047.00 - ₹ 46,191.00 | ||
(2S,5S)-2-tert-Butyl-3-methyl-5-(phenylmethyl)-4-imidazolidinone | -- | ₹ 6,497.00 - ₹ 48,772.00 | ||
346440-54-8 | (2S,5S)-5-Benzyl-2-(tert-butyl)-3-methylimidazolidin-4-one | -- | ₹ 3,204.00 - ₹ 53,667.00 |
Related Products
Description
- General description: (2S,5S)-(-)-2-tert-Butyl-3-methyl-5-benzyl-4-imidazolidinone is a chiral imidazolidinone organocatalyst, developed by MacMillan and co-workers.[1]
- Application: (2S,5S)-(-)-2-tert-Butyl-3-methyl-5-benzyl-4-imidazolidinone is a second-generation MacMillan catalyst, which can be used as a chiral organocatalyst in: The chiral transformation reaction, including Friedel-Crafts and Mukaiyama-Michael reactions.[2] The preparation of substituted spiroundecenetriones via asymmetric domino Knoevenagel/Diels-Alder reactions.[3] The asymmetric synthesis of β-hydroxy aldehydes and their dimethylacetals via aldehyde-aldehyde aldol condensation reaction.[4] The enantioselective α-fluorination of aldehydes using N-fluorobenzenesulfonamide as a fluorinating agent.[5] The stereoselective preparation of (oxomethyl)oxabicyclo[3.2.1]octenones and tricyclic pyrroles via [4+3] cycloaddition of (trialkylsiloxy)pentadienals to furans.[6]
- Features and Benefits: Advantages of MacMillan imidazolidinone organocatalysts:[1]Superior enantiocontrol in numerous transformationsHigh activities at low catalyst loadingsExtraordinary functional group tolerance
- Legal Information: U.S. Pat. 6,369,243 and related patents apply. For research purposes only.
SAFETY INFORMATION
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves