386529
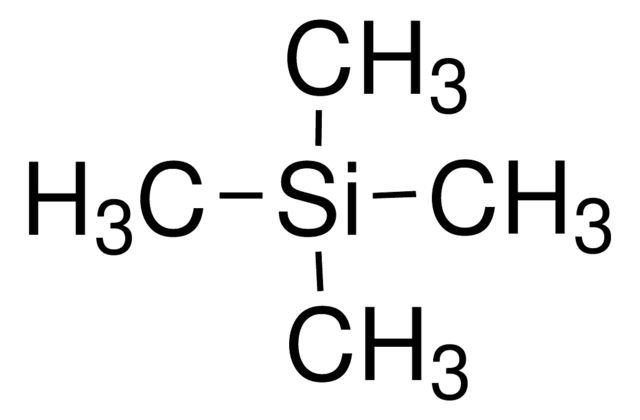
Chlorotrimethylsilane
purified by redistillation, ≥99%
Manufacturer: Sigma Aldrich
CAS Number: 75-77-4
Synonym(S): TMSCl, TMCS, Trimethylchlorosilane, Trimethylsilyl chloride
Select a Size
| Pack Size | SKU | Availability | Price |
|---|---|---|---|
| 25 ML | 386529-25-ML | In Stock | ₹ 3,344.93 |
| 100 ML | 386529-100-ML | In Stock | ₹ 6,765.63 |
| 1 L | 386529-1-L | In Stock | ₹ 37,541.10 |
386529 - 25 ML
In Stock
Quantity
1
Base Price: ₹ 3,344.93
GST (18%): ₹ 602.087
Total Price: ₹ 3,947.017
vapor density
3.7 (vs air)
Quality Level
200
vapor pressure
100 mmHg ( 25 °C)
Assay
≥99%
form
liquid
autoignition temp.
752 °F
purified by
glass distillationredistillation
expl. lim.
6.4 %
impurities
<0.1% dichlorodimethylsilane
refractive index
n20/D 1.387 (lit.)
Other Options
Description
- General description: Chlorotrimethylsilane is a chloroorganosilane compound mainly used for silylation reactions.[1]
- Application: Chlorotrimethylsilane (TMSCl) can be used as: A reagent to protect alcohol and amine groups via the formation of trimethylsilyl ethers and trimethylsilyl amines.[2][3] A catalyst for the preparation of 1,3-diphenyl-2-propenone derivatives (chalcones) as antimicrobial agents.[3] A trapping agent for the anions generated during acyloin condensation reaction.[4] A better alternative catalyst to the toxic mercuric chloride for the activation of samarium (Sm) during the cyclopropanation of both allylic and α-allenic alcohols.[5] A catalyst in the transesterification of triglycerides with alcohols to form fatty acid alkyl esters.[6] A reagent along with lithium bromide for the conversion of alcohols to the corresponding bromides.[7] A reagent in Fischer glycosidation.[8] A source of acid catalyst in the reductive benzylation reaction using benzaldehyde and Et3SiH.[9]A reagent to synthesize sodium trimethylsilanethiolate (Me3SiSNa) by reacting with sodium sulfide, which is an odorless alternative method of synthesizing Me3SiSNa from foul-smelling bis(trimethylsilyl)sulfide and sodium methoxide.[10]
SAFETY INFORMATION
Pictograms
Signal Word
Danger
Hazard Statements
Precautionary Statements
P210 - P233 - P280 - P303 + P361 + P353 - P304 + P340 + P310 - P305 + P351 + P338
Hazard Classifications
Acute Tox. 3 Inhalation - Acute Tox. 3 Oral - Acute Tox. 4 Dermal - Eye Dam. 1 - Flam. Liq. 2 - Skin Corr. 1A
Supplementary Hazards
EUH014
WGK
WGK 1
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves