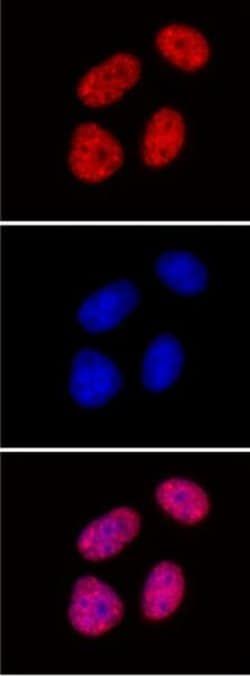
7188674
Histone H3S28ph Monoclonal Antibody (5F9A9), Active Motif™
Mouse Monoclonal Antibody
Manufacturer: Fischer Scientific
The price for this product is unavailable. Please request a quote
Antigen
Histone H3S28ph
Concentration
1 mg/mL
Classification
Monoclonal
Form
Liquid
Regulatory Status
RUO
Formulation
PBS with 30% glycerol and 0.035% sodium azide; pH 7.5
Isotype
IgG2b
Purification Method
Protein A
Product Type
Antibody
Clone
5F9A9
Applications
Immunocytochemistry, Immunofluorescence, Western Blot
Conjugate
Unconjugated
Host Species
Mouse
Target Species
Human
Immunogen
This antibody was raised against a peptide corresponding to phospho-serine 28 of human Histone H3.
Primary or Secondary
Primary
Content And Storage
-20°C, Avoid Freeze/Thaw Cycles
Description
- Description Histone H3 is one of the DNA-binding proteins found in the chromatin of all eukaryotic cells
- H3 along with four core histone proteins binds to DNA forming the structure of the nucleosome
- Histones play a central role in transcription regulation, DNA repair, DNA replication and chromosomal stability
- Post translationally, histones are modified in a variety of ways to either directly change the chromatin structure or allow for the binding of specific transcription factors
- The N-terminal tail of histone H3 protrudes from the globular nucleosome core and can undergo several different types of post-translational modifications that influence cellular processes
- These modifications include the covalent attachment of methyl or acetyl groups to lysine and arginine amino acids and the phosphorylation of serine or threonine.