C3144
Coenzyme A sodium salt hydrate
cofactor for acyl transfer
Manufacturer: Sigma Aldrich
CAS Number: 55672-92-9
Synonym(S): CoA Na2
Select a Size
| Pack Size | SKU | Availability | Price |
|---|---|---|---|
| 10 MG | C3144-10-MG | In Stock | ₹ 7,750.00 |
| 25 MG | C3144-25-MG | In Stock | ₹ 13,670.00 |
| 100 MG | C3144-100-MG | In Stock | ₹ 41,091.70 |
| 500 MG | C3144-500-MG | In Stock | ₹ 1,42,803.40 |
| 1 G | C3144-1-G | In Stock | ₹ 2,57,819.03 |
C3144 - 10 MG
In Stock
Quantity
1
Base Price: ₹ 7,750.00
GST (18%): ₹ 1,395.00
Total Price: ₹ 9,145.00
description
cofactor for acyl transfer
Quality Level
200
Assay
≥85% (spectrophotometric assay)
form
powder
solubility
H2O: soluble 50 mg/mL, clear, colorless to faintly yellow
storage temp.
−20°C
SMILES string
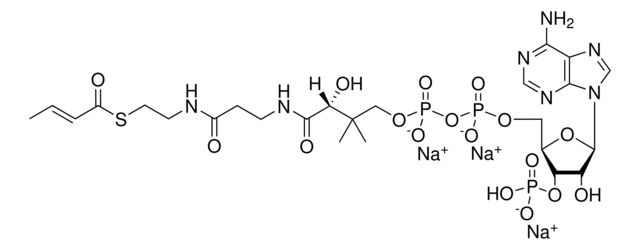
[Na+].CC(C)(COP(O)(=O)OP(O)(=O)OC[C@H]1O[C@H]([C@H](O)[C@@H]1OP(O)([O-])=O)n2cnc3c(N)ncnc23)C(O)C(=O)NCCC(=O)NCCS
InChI key
SYTRWOCXZXQBPW-CLVRNSBASA-M
Other Options
| Image | Product Name | Manufacturer | Price Range | |
|---|---|---|---|---|
Sigma Aldrich Fine Chemicals Biosciences Coenzyme A sodium salt hydrate cofactor for acyl transfer | 55672-92-9 (anhydrous) | 1G | -- | ₹ 2,67,507.30 | ||
Sigma Aldrich Fine Chemicals Biosciences Coenzyme A Sodium Salt Hydrate | 55672-92-9 (anhydrous) | 25mg | -- | ₹ 16,571.80 | ||
Sigma Aldrich Fine Chemicals Biosciences Coenzyme A sodium salt hydrate suitable for cell culture, BioReagent | 55672-92-9 (anhydrous) | 100MG | -- | ₹ 48,166.80 | ||
Coenzyme A (sodium) | -- | ₹ 1,02,528.00 | ||
55672-92-9 | Coenzyme a sodium salt hydrate | -- | ₹ 2,937.00 - ₹ 12,460.00 |
Related Products
Description
- General description: Coenzyme A (CoA) is an essential cofactor in living systems and is synthesized from pantothenic acid (vitamin B5),[1] The CoA levels in mitochondria and peroxisomes correspond to 2-5 mM and 0.7 mM, respectively. Cytosolic CoA is in the range of 0.05 mM to 0.14 mM[1]
- Application: Coenzyme A is suitable for use in:gylcerolipid biosynthesis in porcine adipose tissue[2]an assay to measure the level of Alpha-methylacyl-CoA racemase (AMACR) in human blood samples using a nanoparticle electrochemical biosensor[3]chloramphenicol acetyltransferase (CAT) assay[4][5]the synthesis of palmitoyl-CoA, which is required for palmitoylation and activation of proteins for regulated membrane fusion[6]
- Biochem/physiol Actions: Coenzyme A (CoA, CoASH, HSCoA) is a coenzyme that facilitates enzymatic acyl-group transfer reactions and supports the synthesis and oxidation of fatty acids. CoA is involved in the mechanisms of a wide variety of enzymes. In the presence of CoASH, organic carboxylic acids form acyl-CoA thioesters, which facilitates enzyme recognition. The acyl-CoA formed from xenobiotic carboxylic acids can add to the compound′s toxicity, which can lead to cellular metabolic dysfunction.[7] It is involved in the oxidation of pyruvate in the Kreb′s cycle. CoA is needed for metabolic events.[8] The bacterial CoA pathway is targeted for antimicrobial development.[8] It mediates acyl group transfer and carbonyl activation. The CoA and its thioester levels are crucial for cellular homeostasis. CoA is also involved in regulating platelet aggregation and vasoconstriction.[1] It acts as an essential cofactor in enzymatic acetyl transfer reactions.
SAFETY INFORMATION
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves